Life Science Market

In 2020 ATC reshaped the organization and established a Life Science Division to focus 20 years of experience to a separate business unit. This has allowed ATC to better embrace the demands and rigors of Automated Assembly for Medical Device, Pharmaceutical & Healthcare Industry. All systems are custom designed to meet the User Requirement Specifications (URS), leveraging some of the most comprehensive collection of proprietary technologies and delivering best in class results.
While there are areas of core competency, ATC Automation leverages the automation platforms appropriate to meet the demands of each customer’s URS. From Asynchronous Motion Assembly, Indexing Technology, Servo motion, Robotic assembly to fixed process, and data collection demands, ATC has the necessary flexibility to create an assembly solution that meets the needs for market ROI of every size and scope.
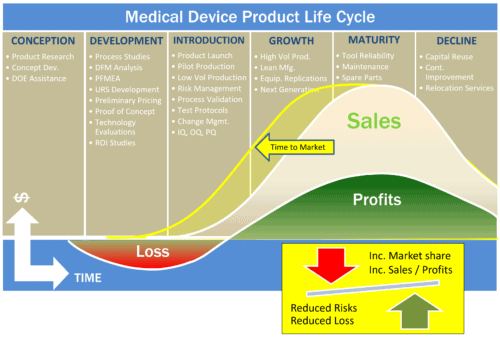
ATC dedicated project teams are focused not just on delivering world class results, but the team embraces and understands that the machine built for you has be completed with validation in mind and a clear protocol execution. ATC continues to advance every aspect of the assembly process whether it leverages past experience or requires a new proof of principle to ensure that only winning solutions are leveraged at the finish line. This is a risk averse methodology that leads to solutions that exceed our customers requirements. While all of the process are too extensive to mention by individual process, it makes sense to touch on just a few.
Below are a several specific product families and processes:
- Valves
- Catheters
- Pumps
- Electric Motors
- Pharma Delivery Devices
- Battery Cells and Modules
- Epi Pens (Autoinjector)
- Robotic Assembly
- Micro Motor & Assemblies
- Syringe Assembly
- Needle Handling & Inspection
- Tray Handling
- Smart Devices
- Bluetooth Enabled Devices
- Prefilled Devices
- Surgical Device Assembly
- Tubing Processing and Assembly
- Safety Device Assembly
- Diagnostic Device Assembly
- Blood Collection Devices
- Blades and specialty devices
- Auto Injectors
- Oxygen Devices
- Dental Devices
- Implants
- Powered Surgical Tools
- Dispensing Solutions
- Ultrasonic Assemblies
- One Way Values
- Test Strips
- Ocular Injection
- Ventilators
While in no way does this represent all of the systems ATC has delivered, it touches many of these products that require discrete part assembly in Robotic automation, fixed automation, and the verification systems that ensures safety and complete part traceability.
Automation can be deployed to fasten, snap, rivet, attach, and inspect by machine vision by being integrated with Robotic cells or fixed automation. Smart devices are getting better and with more sensed motion for reliability, speed, feedback, and assurance the optimization in assembly can be assured.
Robotic Assembly
Medical assembly are all unique solutions that require the rigors of product handling and quality inspection. ATC integrates Cobots, Catesian, Scara, and multi Axis Robots into many work cells and automation solutions as they are required. With the vast experience at our disposal integrating simple and complex vision into our systems ensures precise placement and feedback of all process when utilizing Robotic Solution.

Indexed Motion
These types of platforms are used when an assembly or inspection task requires a fixed cycle time. These devices are readily available and carry robust mechanisms at a fairly high precision. +/- 0.04mm or +/- 0.05mm accuracy on index and consistent part to part repeatability in a variety of size and dimensions.
Clean Room
Clean Rooms are expensive and the space is also very costly to our customers. ATC manages every opportunity the minimize foot print while keeping access safe and reasonable. ATC Automation’s equipment is designed to your specific requirements but has been completed for the operating requirements as stringent as ISO 4, to more traditional of Class 7 and Class 8.
Often times clean rooms production requires lubricants meet necessary FDA classifications, and all materials that are in contact with the product must be approved for use in drug manufacturing equipment. All surfaces that contact components are not reactive additive or absorptive. When required, Laminar air flow is managed regularly with additional filtering and air movement to minimize any contamination.
Diagnostic Device Assembly
Diagnostic devices have evolved and are no longer simple strips. Today’s devices leverage smart technology and complex disposables all in the name of improved accuracy and efficacy. ATC has deployed several high speed solutions to deliver reliable assembly at a rate the market demands. These can be life enhancers for micro components.
Surgical Device
ATC has automated a wide range of surgical devices to meet the demands of todays surgeons. While the product development is not always optimized for assembly and testing, ATC leverages its experiences to meet the demands of assembling the smallest components in a safe and reliable method. 100% of the time these assemblies must be perfect as they are often times dealing with catastrophic outcomes if failure occurs. It is with this mindset, we deliver mission critical devices to the end customer with the very best reliability and assurance across wide assembly platforms that generally have feedback loop on every assembly operation.
Validation
The job is never done until the paperwork is done and rest assured ATC is leveraging Risk Based methodology to ensure each piece of equipment can be validated to the IQ/OQ/PQ protocols necessary to meet GMP requirements. ATC has in house a validation department that customizes the necessary approach and plan needed for each customer’s unique requirements.
The above represent just a few of the specifics ATC has automated. When getting to market is essential and patented products are on the clock, fast efficient execution will make a difference as to ROI and life cycle performance. Call ATC today to explore some of the assembly solutions that can make a difference in your specific life science niche.